Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. These concepts form the basis of chemistry. 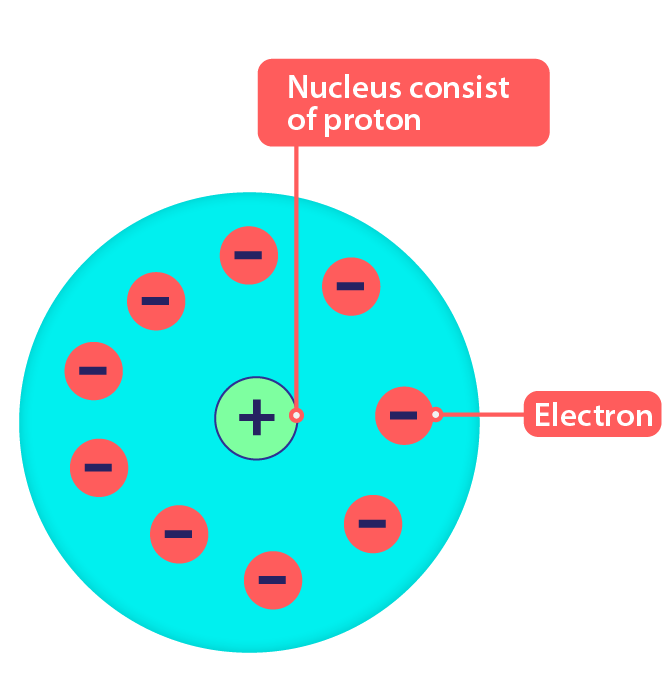
Atoms combine in whole-number ratios to form compounds. An element consists of only one type of atom, which has a mass that is characteristic of the element and is the same for all atoms of that element. An atom is the smallest unit of an element that can participate in a chemical change. Atoms of the same element are the same atoms of different elements are different. Here are the postulates of Dalton’s atomic theory: Matter is composed of exceedingly small particles called atoms. His model postulated that atoms consist of a big positively-charged sphere studded with negatively charged electrons (he called them 'corpuscles') like fruit in a plum pudding. 
It consists of three parts: All matter is composed of atoms. Thomson proposed the 'plum pudding' theory of the divisible atom in 1904, after discovering electrons in 1897. Scientists did not account for the gases that play a critical role in this reaction. , first stated by John Dalton, an English scientist, in 1808. \) shows that the burning of word does follow the law of conservation of mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
